|
The effect decreases when one travels further to the right in a time due to repulsions from the electron-electron which otherwise would cause the size of the atom to increase. For facts, physical properties, chemical properties, structure and atomic properties of the specific element, click on the element symbol in the below periodic table. 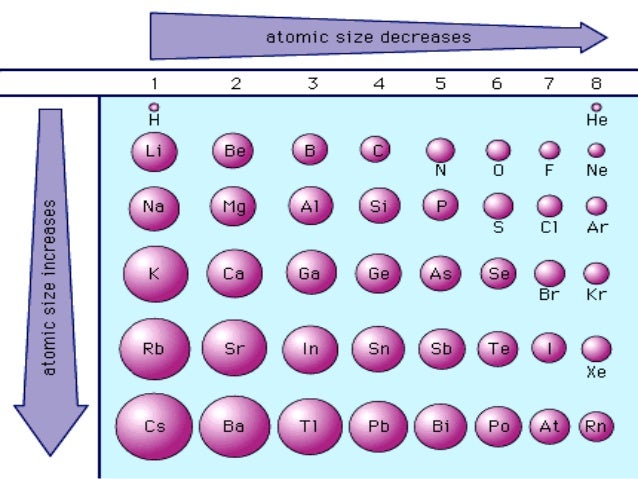
In the below periodic table you can see the trend of Atomic Radius. Fluorine, 42 pm, Chromium, 166 pm, Rubidium, 265 pm.Text lists sorted by: Value Atomic Number Al. Periodic Table of Elements with Atomic Radius Trends. Neon, 38 pm, Silver, 165 pm, Barium, 253 pm. NOTE: Option B, C, D is wrong because the elements of 4th and 5th period are larger in size of atomic radius as compared to period 3rd and helium which is a noble gas and hydrogen with only one electron in its shell, as the force of attraction increases between nuclei and electrons the size of the atoms decreases. Arrange the following in order of decreasing atomic radius: Pt, At, Ba, Sm Which element would you predict to have the highest ionization energy Get Document So, in general, ionization energy increases from the bottom-left to the upper-right of the periodic table Oganesson: Value is a guess based on periodic table trend The atomic radius of. Helium, 31 pm, Cadmium, 161 pm, Praseodymium, 247 pm. Hence, Option A is correct which is I >Br >S >He >H. Na has largest size among these because according to the trend, atomic radius decreases as we move from left to right in a period of the periodic table. Therefore the decreasing order of atomic radius is I >Br >S >He >H So, according to this concept, I and Br belong to the halogen family of the 4th and 5th period so they are larger in atomic size, S belongs to the 3rd period and group 16, He is a noble gas with two electrons and similar in size but H with one electron is of least atomic size. Therefore the radius of atoms increases as you go down a certain group in the periodic table of elements.Īs per for the periods, when we move left to right in a particular period the size of an atom decreases. Asked by Topperlearning User 4th Jun, 2014, 01:23: PM Decreasing order of atomic size: Li > Be > B > C > N > O Answered by 4th Jun, 2014, 03:23: PM.  
decreases or increases from.Īs we know, that an atom gets larger as it’s the number of shells increases. Hint: To solve these types of questions you should know about the properties of groups and periodic table periods, e.g.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
